![[Left] Man finding a lucky penny. [Right] Man contemplating the luck a penny could bring him.](https://usim.beprod.ilarishcp.com/sites/ilarishcp_com/files/styles/hero_full_width_width_2560/public/2024-08/mkd-hero-image-d.png?itok=5Ywu4cGD)
Tumor Necrosis Factor Receptor–Associated Periodic Syndrome (TRAPS)
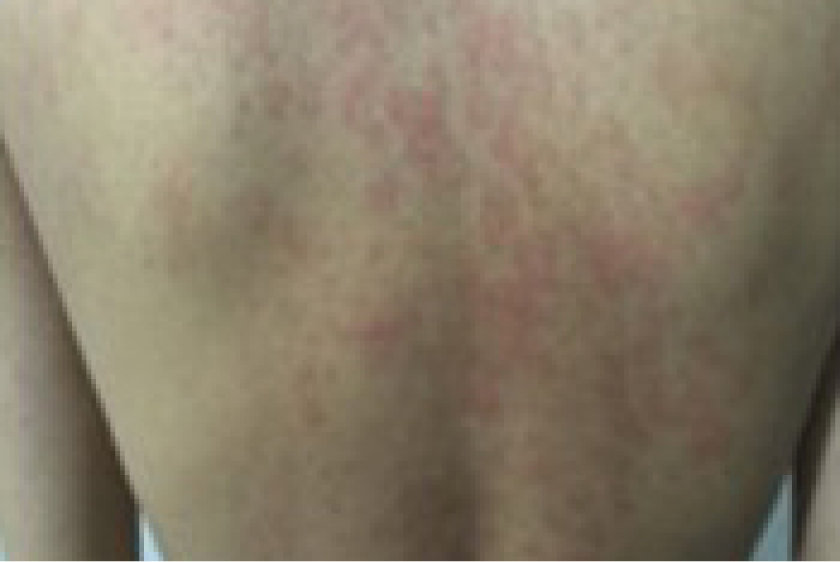
Not an actual patient. Individual results will vary.
TRAPS is hereditary and can emerge from early childhood to adulthood1-9
| Attribute | Details |
| Cutaneous findings |
|
| Predominant ethnic distribution | All ethnicities |
| Worldwide prevalence or number of cases | >1000 |
| Typical age at onset | Varies; <3 years to <20 years |
| Duration of attacks | 7 to 28 days; nearly continuous in one-third of patients |
| Frequency of attacks | Irregular; 5 weeks to months or years |
| Gene mutation | TNFRSF1A |
| Inheritance | Autosomal dominant |
| Other select clinical features |
|
| High serology | Increase in CRP, ESR, and SAA |
Rash image credit: Reproduced with permission from Kosar Asnaashari and Nima Rezaei, Chapter 28 - Rheumatologic manifestations of autoinflammatory diseases, 2023, page 575, figure 3.
References: 1. Hoffman HM, Simon A. Recurrent febrile syndromes—what a rheumatologist needs to know. Nat Rev Rheumatol. 2009;5(5):249-256. doi:10.1038/nrrheum.2009.40 2. Kastner DL. Hereditary periodic fever syndromes. Hematology Am Soc Hematol Educ Program. 2005;(1):74-81. doi:10.1182/asheducation-2005.1.74 3. Genetics Home Reference. Tumor necrosis factor receptor-associated periodic syndrome. US National Library of Medicine; 2020. Accessed June 9, 2024. http://ghr.nlm.nih.gov/condition/tumor-necrosis-factor-receptor-associated-periodic-syndrome 4. Lachmann HJ, Hawkins PN. Developments in the scientific and clinical understanding of autoinflammatory disorders. Arthritis Res Ther. 2009;11(1):212. doi:10.1186/ar2579 5. Hausmann JS, Dedeoglu F. Autoinflammatory diseases in pediatrics. Dermatol Clin. 2013;31(3):481-494. doi:10.1016/j.det.2013.04.003 6. Kimberley FC, Lobito AA, Siegel RM, Screaton GR. Falling into TRAPS—receptor misfolding in the TNF receptor 1-associated periodic fever syndrome. Arthritis Res Ther. 2007;9(4):217. doi:10.1186/ar2197 7. Ciccarelli F, De Martinis M, Ginaldi L. An update on autoinflammatory diseases. Curr Med Chem. 2014;21(3):261-269. doi:10.2174/09298673113206660303 8. Jesus AA, Oliveira JB, Hilário MO, et al. Pediatric hereditary autoinflammatory syndromes. J Pediatr (Rio J). 2010;86(5):353-366. doi:10.2223/JPED.2015 9. Barron KS, Kastner DL. Periodic fever syndromes and other inherited autoinflammatory diseases. In: Petty RE, Laxer RM, Lindsley CB, Wedderburn LR, eds. Textbook of Pediatric Rheumatology. 7th ed. Elsevier; 2016:609-626.